Here is a list of the most important methods used in the NIC:
Electroencephalography (EEG) is the most commonly used method to non-invasively monitor neuronal activity in the brain via surface electrodes attached to the scalp (up to 64 or 128 in an EEG-cap), measuring and amplifying the voltage difference between different electrode sites. The weak EEG potential at the scalp (in microvolts) depends on the summation of extracellular synchronized postsynaptic currents of large neuron populations, producing a polarization of ions in the brain tissue outside the neurons (the local field potential, LFP), Since the EEG signal measured at each electrode reflects a mixture of numerous active sources in the brain, source localization methods are required to make inferences about the local origin of specific EEG phenomena. EEG can be analyzed with respect to the frequency, power, phase, or spatial coherency of spontaneously occurring or experimentally induced oscillations, or the amplitude and latency of certain event-related (evoked) potentials. Our lab uses EEG to study neuronal oscillations during wakefulness and sleep, and to quantify the cortical response to transcranial brain stimulation.
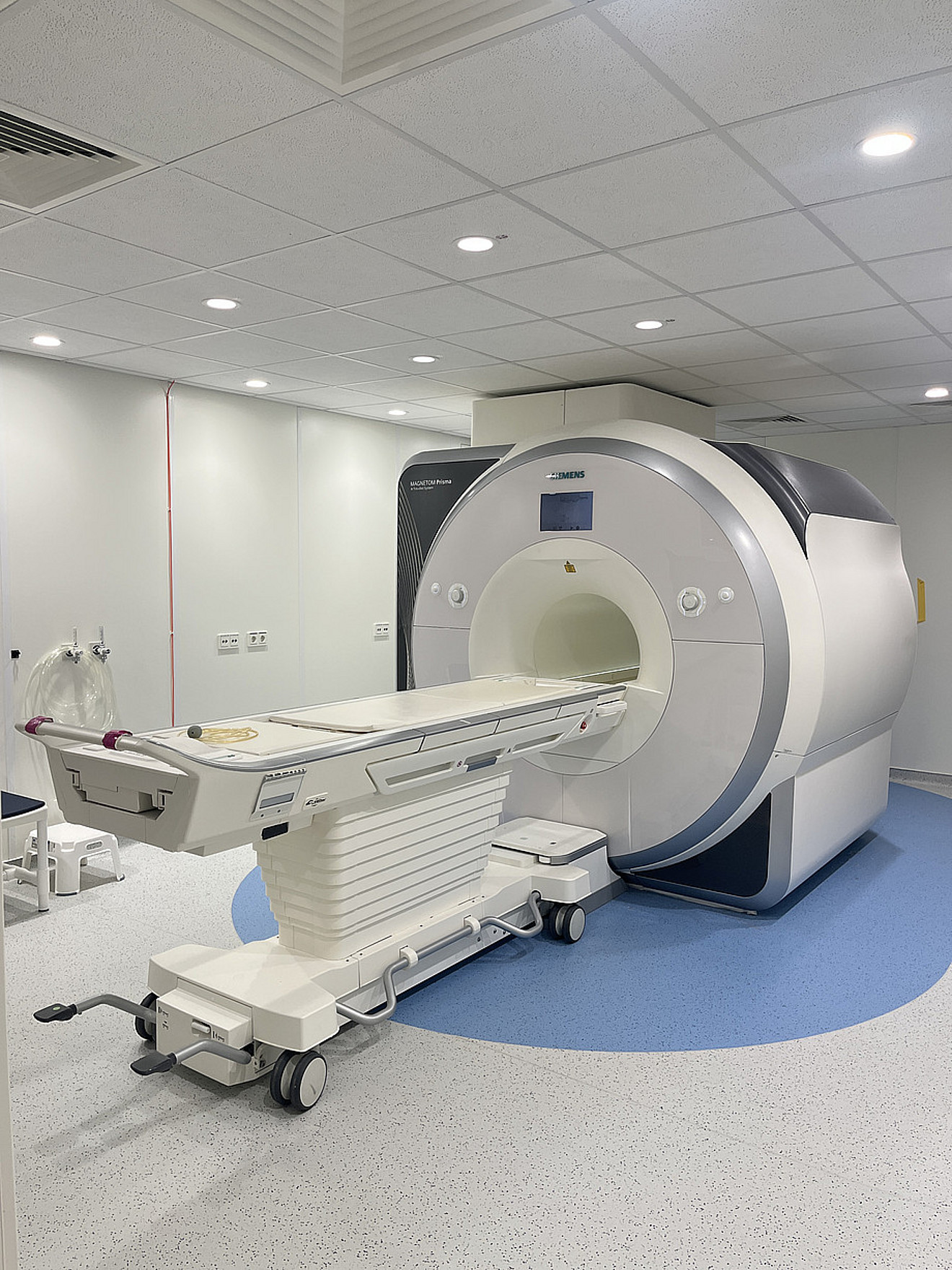
Magnetic Resonance Imaging (MRI) is a non-invasive imaging technique that uses strong magnetic fields and radiofrequency pulses to generate high-resolution images of the body, particularly the brain. Unlike X-ray or CT imaging, MRI does not involve ionizing radiation. The technique is based on detecting signals from hydrogen nuclei in tissue water and fat, which vary depending on the molecular environment. These signal differences are reconstructed into detailed anatomical or functional images. MRI therefore provides versatile tools for investigating brain structure, physiology, and function in vivo. Although our primary focus is on functional MRI, multiple MRI sequences are available to address a broad range of research questions. The most important MRI sequences used at the NIC (in alphabetical order) are:
Arterial Spin Labeling (ASL)
ASL is a perfusion MRI technique that quantifies cerebral blood flow by magnetically labeling inflowing arterial blood water as an endogenous tracer. By comparing labeled and control images, regional perfusion can be quantified non-invasively. In contrast to BOLD-fMRI, ASL provides an absolute measure of blood flow rather than a relative oxygenation signal. It is particularly useful in studies of cerebrovascular function, aging, and neurological disorders affecting cerebral perfusion.
Diffusion Weighted Imaging (DWI)
DWI measures the diffusion of water molecules within brain tissue. In white matter, diffusion is directionally constrained along axonal fibers, enabling the characterization of microstructural organization. Diffusion Tensor Imaging (DTI), a common extension of DWI, models diffusion directionality and enables reconstruction of white matter tracts. DWI is widely used to investigate structural connectivity and is highly sensitive to acute ischemic stroke.
Functional MRI (fMRI)
fMRI assesses brain activity indirectly by measuring changes in blood oxygenation, known as the blood oxygenation level-dependent (BOLD) signal. Neural activation triggers localized changes in cerebral blood flow and oxygenation through neurovascular coupling. These hemodynamic changes are detected as signal fluctuations over time. fMRI can be used to study task-evoked brain responses as well as intrinsic functional connectivity during resting state.
T1- and T2-weighted imaging
T1- and T2-weighted sequences provide high-resolution structural images of brain anatomy. T1-weighted images offer strong gray–white matter contrast and are commonly used for morphometric analyses and anatomical localization. T2-weighted images are sensitive to tissue water content and are particularly useful for detecting edema, inflammation, and lesions.
Polysomnography (PSG) refers to the recording of sleep based on the concurrent measurements of brain waves (electroencephalography, EEG), muscle tone (electromyography, EMG), and eye movements (electrooculography, EOG). Based on these measurements, different sleep stages can be scored: non-rapid eye movement (NREM) and rapid eye movement (REM) sleep as well as sub-stages (N1, N2, N3 as well as phasic and tonic REM sleep). Out lab uses PSG as a standard method to score sleep stages before starting more detailed analyses of certain oscillatory events, such as NREM slow oscillations (< 1Hz) and sleep spindles (~10-16 Hz), or REM delta (~2-4 Hz), theta (~4-8 Hz) and beta (~17-30 Hz).
The NIC operates two fully equipped psychophysiology laboratories, comprising two and three shielded workstations (cubicles), respectively, for conducting experiments without neuroimaging or brain stimulation. Each cubicle is equipped with a monitor and headphones for the presentation of visual and auditory stimuli, as well as a keyboard, joystick, and response keyboard. All workstations allow for the acquisition of psychophysiological measures, including electrocardiography (ECG), skin conductance (electrodermal activity, EDA), and electromyography (EMG).
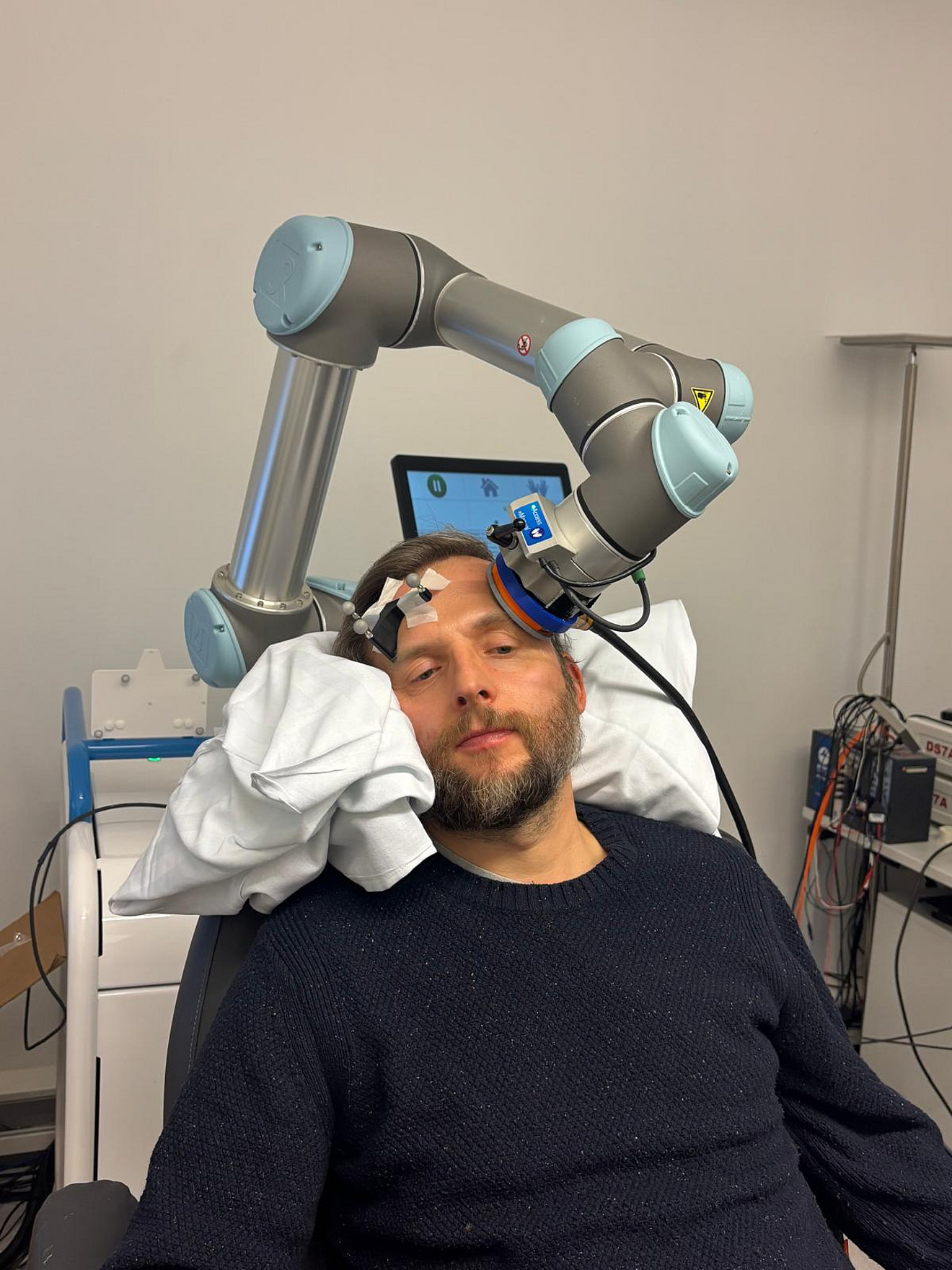
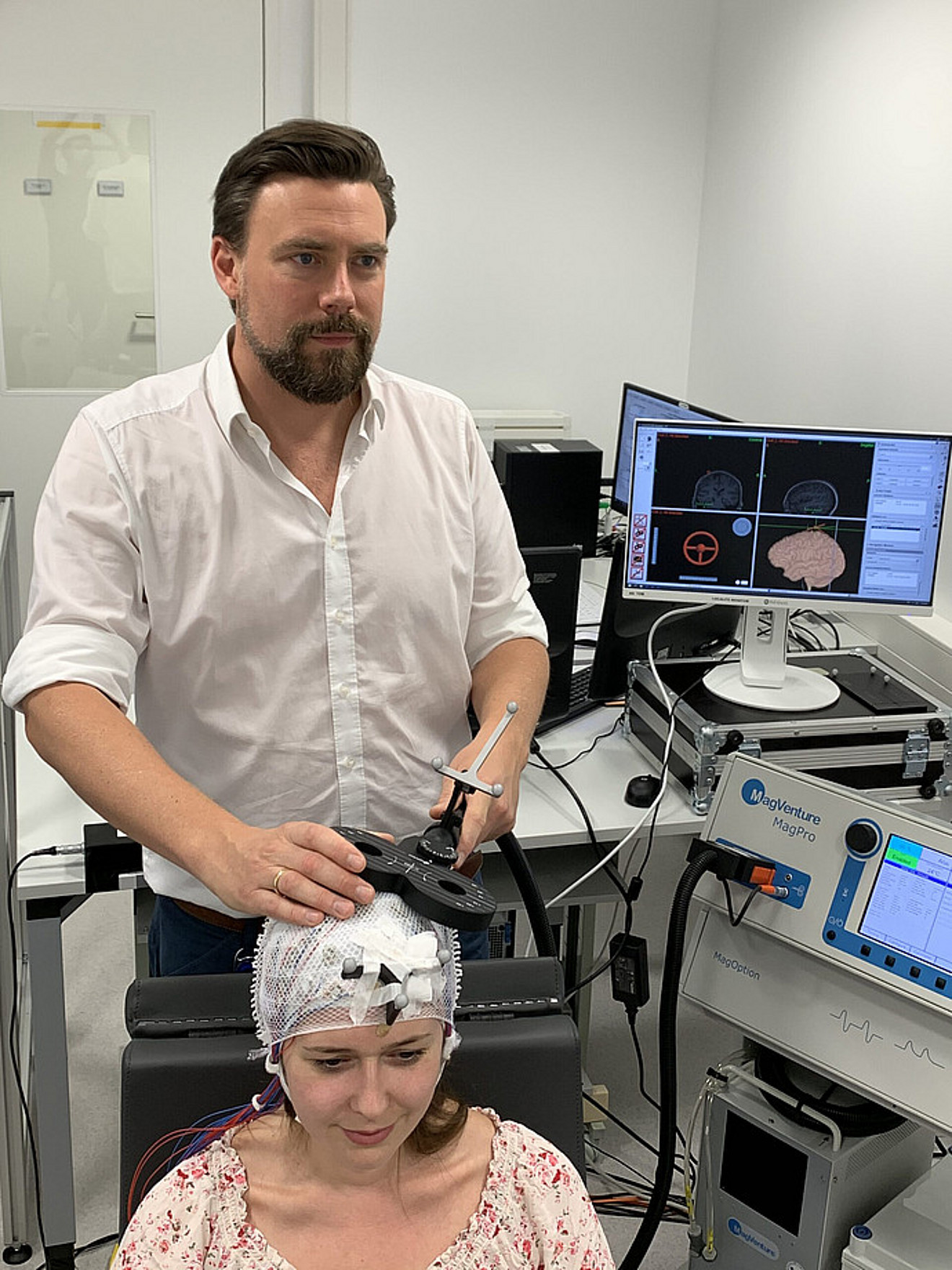
Frameless stereotactic neuronavigation allows to co-register the participant’s head to their structural MR image with the help of a stereo infrared camera and so called trackers (unique geometries made of three infrared-reflecting spheres) being attached the head as well as to a pointer with which multiple strategic points on the surface of the participants head are sampled and automatically aligned with the rendered surface of the structural MRI. Additional trackers on the TMS coil or TUS transducer then allow to navigate these relative to the participants brain with millimetre precision and to bring them into the desired position for stimulating the anatomical target structure. While this MR-based neuronavigation can be conducted manually, robot-assisted navigation has become feasible and will likely become the state-of-the-art in the near future. Robotized neuronavigation allows to automatically bring the TMS coil or TUS transducer into the desired position with the help of a robotic arm, to compensate participant’s head movement in real-time, and to target multiple brain regions in subsequent trials, allowing effective mapping of entire target regions or the experimental randomization of target conditions. If integrated with a powerful experimental control software (e.g., our BEST toolbox in section 4.2), robotized neuronavigation also allows to establish automated motor hotspot searches in a closed-loop setup.
Transcranial Magnetic Stimulation (TMS) is a non-invasive method to stimulate small regions of the brain based on the principle of electromagnetic induction. A magnetic field generator (the TMS coil) is placed on the participant’s head, and for the fraction of a millisecond a strong electric current is discharged through the coil, producing a magnetic field that painlessly reaches into the brain and induces an electric current in the brain tissue, which depolarizes axons and triggers action potentials in cortical neurons. Based on an MR-guided neuronavigation system, the coil can be positioned very precisely to stimulate a circumscribed part of the cortex (and its connected regions). TMS can be applied as single-pulse, paired-pulse, or in bursts for online approaches, and in form of repetitive TMS (rTMS) for offline approaches, and its precise effects depend on the coil location and orientation, as well as the intensity, frequency, and temporal pattern of the delivered magnetic pulses. For example, single-pulse TMS of the primary motor cortex (M1) can evoke twitches in the contralateral muscles, which can be quantified by the amplitude of the motor evoked potential (MEP) in the surface electromyogram (EMG) and indicates corticospinal excitability. Repeated MEP measurements can indicate changes in corticospinal excitability due to spontaneous changes in in brain state or experimental manipulations (e.g., rTMS). Our lab is using TMS mainly in combination with neuroimaging techniques (see below) to measure excitability and connectivity, entrain neuronal oscillations, and induce transient excitability changes.
Transcranial ultrasonic stimulation (TUS) is a newly emerging technique for non-invasive neuromodulation in humans, using ultrasonic waves instead of electric fields to interact with neural targets in the brain. Special ultrasound transducers, placed on the participants head, transmit a beam of low-intensity ultrasonic waves through the intact human skull, which can be steered precisely and focused on a few cubic millimetres of brain tissue at several centimetre distance. TUS thereby allows for the first time to non-invasively, reversibly, and exclusively target with unprecedented precision also very small subcortical brain structures, without co-stimulating more superficial cortical regions (which is impossible with TMS and tES). TUS comes with (at least) two key innovations that will likely revolutionize non-invasive neurostimulation in both fundamental research and psychiatry within the next decade: Firstly, TUS allows the direct modulation of local neuronal activity (presumably via the interaction with mechanosensitive ion channels and possibly with the neurons’ membrane itself), with specific protocols being able to induce also plasticity-related after-effects. Secondly, when combined with the injection of microbubbles into the blood stream, TUS is able to transiently and locally open the blood-brain barrier (BBB), thereby enabling the focal uptake of pharmaceutical (or other active) compounds otherwise unable to pass the BBB, providing a mechanism for targeted drug delivery. Current developments in TUS transducer technology will soon allow to flexibly control the focus and shape of the ultrasound beam, enabling multi-focal and moving stimulation patterns adjusted to individual neuroanatomy and specific brain functions. Importantly, the above described applications use low-intensity focused ultrasound (LIFU) only, and the device (NeuroFUS, Brainbox Ltd, UK) and protocols we use do not produce relevant temperature increases in the brain tissue. LIFU for neuromodulation or BBB opening has thus to be distinguished from the high-intensity focused ultrasound (HIFU) protocols produced by neurosurgical devices, which are explicitly used for the non-invasive thermal ablation of brain tissue, e.g., for the treatment of tremor in Parkinson’s disease.
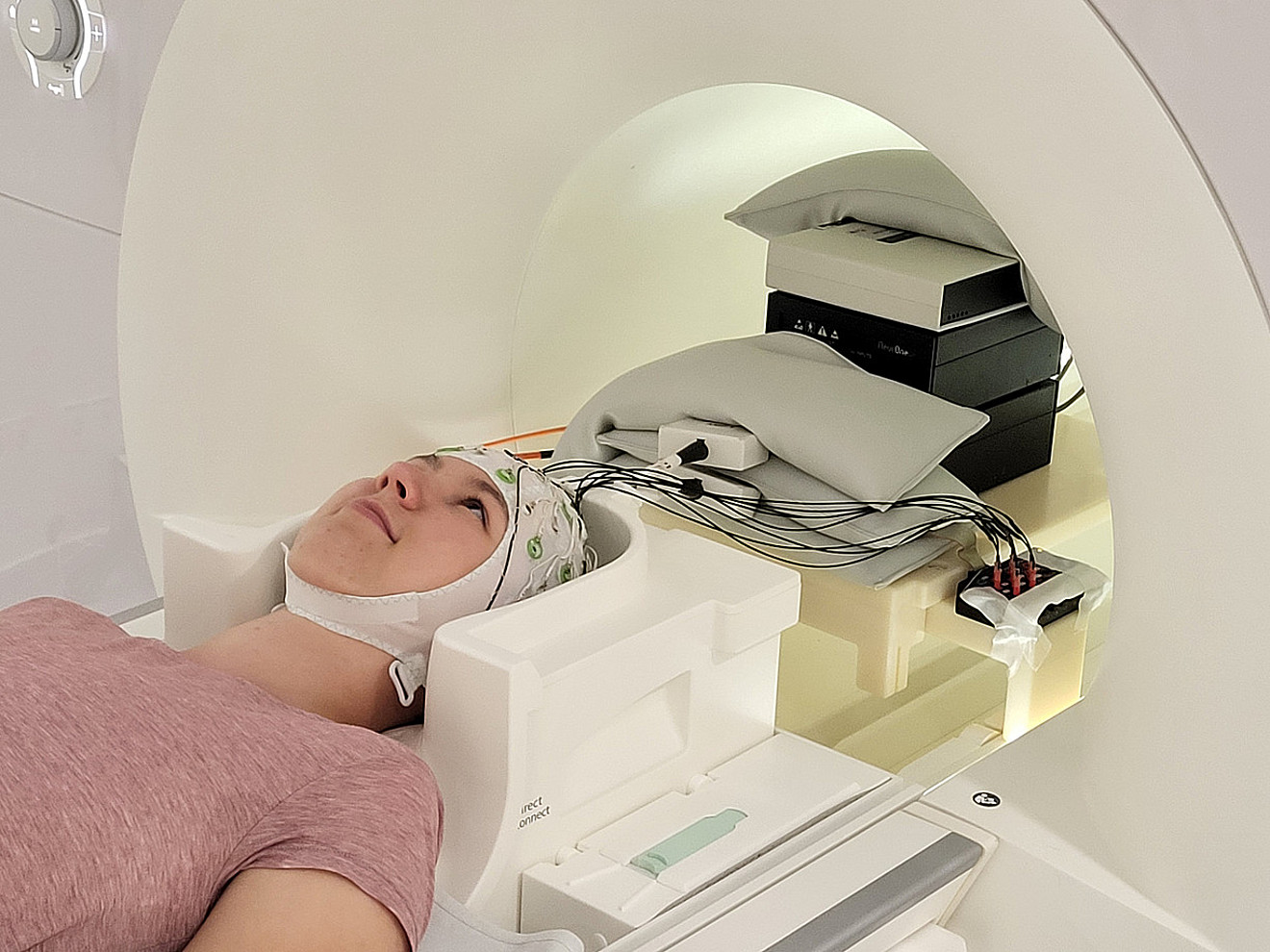
The concurrent recording of EEG and fMRI allows to co-register the complementing information from both techniques, taking advantage of the high temporal resolution of EEG and the high spatial resolution of fMRI. Given the large magnetic fields and radiofrequency pulses in the MR-environment and the artifacts caused in the EEG due to the magnetic gradients of fMRI sequences and heart-beat related head/electrode movements in the static magnetic field of the MRI, special MR-compatible EEG equipment, specific recording procedures, and sophisticated EEG-artifact correction methods are required to gain high quality EEG-fMRI data. However, their combination is worthwhile when EEG and fMRI data from the same trials or spontaneous events needs to be related. Our group employs concurrent EEG-fMRI to study the brain networks and neuronal activations in subcortical brain structures that are associated with certain oscillatory events in wakefulness and during sleep.
On the one hand, concurrent TMS-EEG can either be used to measure the direct brain response to TMS, the TMS-evoked EEG potential (TEP) or TMS-induced oscillations (TIO), in regions outside the primary motor cortex (M1), where motor-evoked potentials (MEP) cannot be assessed. TEPs can be utilized to index cortical excitability, effective connectivity, and the general complexity and spectral composition of a brain response. TMS causes a number of complex artifacts in the EEG, resulting from magneto-electric induction, electrode/electrolyte depolarization, vibration, cranial muscle activation, etc. that need to be attenuated during the recording and removed during pre-processing before EEG analysis (respective pipelines exist for FieldTrip and EEGlab). However, TMS is inevitably associated with sensory-co-stimulation effects from auditory (the TMS ‘click’ sound) and somatosensory input (excitation of peripheral and cranial nerves, and cranial muscle twitches) resulting in peripherally evoked potentials (PEPs) that need to be attenuated and actively controlled for. On the other hand, concurrent EEG-TMS can be used to inform the timing (and other stimulation parameters) for real-time EEG-triggered TMS (brain state-dependent brain stimulation, see below), e.g. to study neuronal oscillations.
Concurrent TMS-fMRI allows to use the TMS-evoked BOLD-signal to assess the neuronal response of the brain to transcranial cortical stimulation, revealing both the local response at the stimulation site itself and the network response in remote connected regions, including deep subcortical structures. Beside the assessment of brain networks and effective connectivity, TMS-fMRI can provide ‘proof of target engagement’, that is empirical evidence for having effectively stimulated the targeted brain region. This is particularly relevant when the actual target site is a deep structure that cannot be directly accessed by TMS, and therefore requiring the transsynaptic stimulation via projections from a more superficial cortical entry point. Measuring the direct BOLD-response to stimulation also allows to determine the optimal stimulation parameters (coil orientation, intensity, frequency, etc.) for a certain target region, going beyond the mere referencing to the individual resting motor threshold (RMT) even for non-motor regions at different scalp-cortex distance and orientation. Our lab is currently establishing TMS-fMRI to optimize stimulation parameters for both superficial and deep target structures, with the aim to demonstrably stimulate memory- and emotion-related deep brain structures and to investigate their role in fear memory consolidation and emotional memory updating in the context of resilience.