Research Group Skevaki – Virus immunology, chronic inflammation and enviromental Health
Background
Our research aims to
- better understand virus-driven immune endotypes in chronic diseases in order to develop immunotherapeutic approaches,
- identify robust biomarkers for precision diagnostics, and
- investigate how environmental pollutants influence immune responses, the course of infections, and the development of chronic diseases.
Current research projects in our group focus on biomarkers for the endotyping of Long COVID, T-cell exhaustion in Crimean-Congo hemorrhagic fever virus (CCHFV) infection in the context of immune responses in COPD and chronic viral infections, respiratory virus-associated asthma and COPD, and the effects of wood smoke exposure on cellular immunity, among other topics. In addition, we conduct epidemiological studies focusing on the effects of environmental exposures (e.g., solar radiation, ionizing radiation) on immunological biomarkers as well as infections.
Projects
Project 1
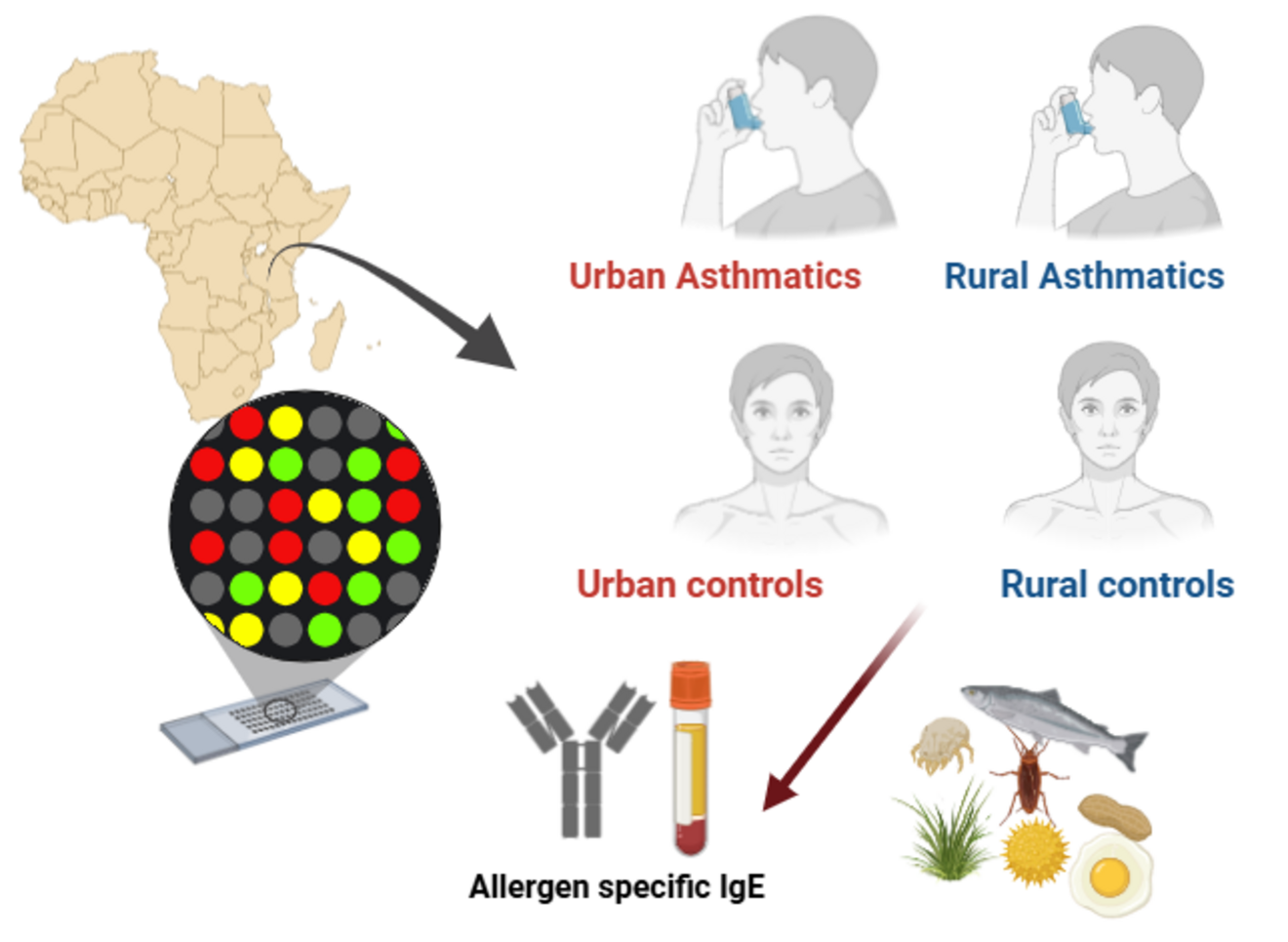
Allergen-Specific IgE Sensitization Profiles and the Role of Virus Infections among Asthmatic Adults in Sub-Saharan Africa (The RIPA2TAN Study)
Sub-Saharan Africa (SSA) is experiencing rapid economic growth and urbanization, accompanied by a marked increase in the prevalence and incidence of non-communicable diseases (NCDs). Reported prevalence of allergy-related symptoms varies widely across the region, ranging from approximately 3% to 27%. Asthma and atopic sensitization have been reported to be more common in urban populations compared with rural populations in SSA. We hypothesize that the differences in environmental and microbial exposures, as well as living conditions between urban and rural asthmatic patients, influence the immunological and molecular endotypes of asthma.
The aims of this project:
- To characterize the IgE sensitization profile of adult asthmatic patients in SSA.
- To compare IgE sensitization patterns between urban and rural populations.
- To construct a multilayer network based on allergen-specific IgE data to better understand patterns of sensitization.
Project 2
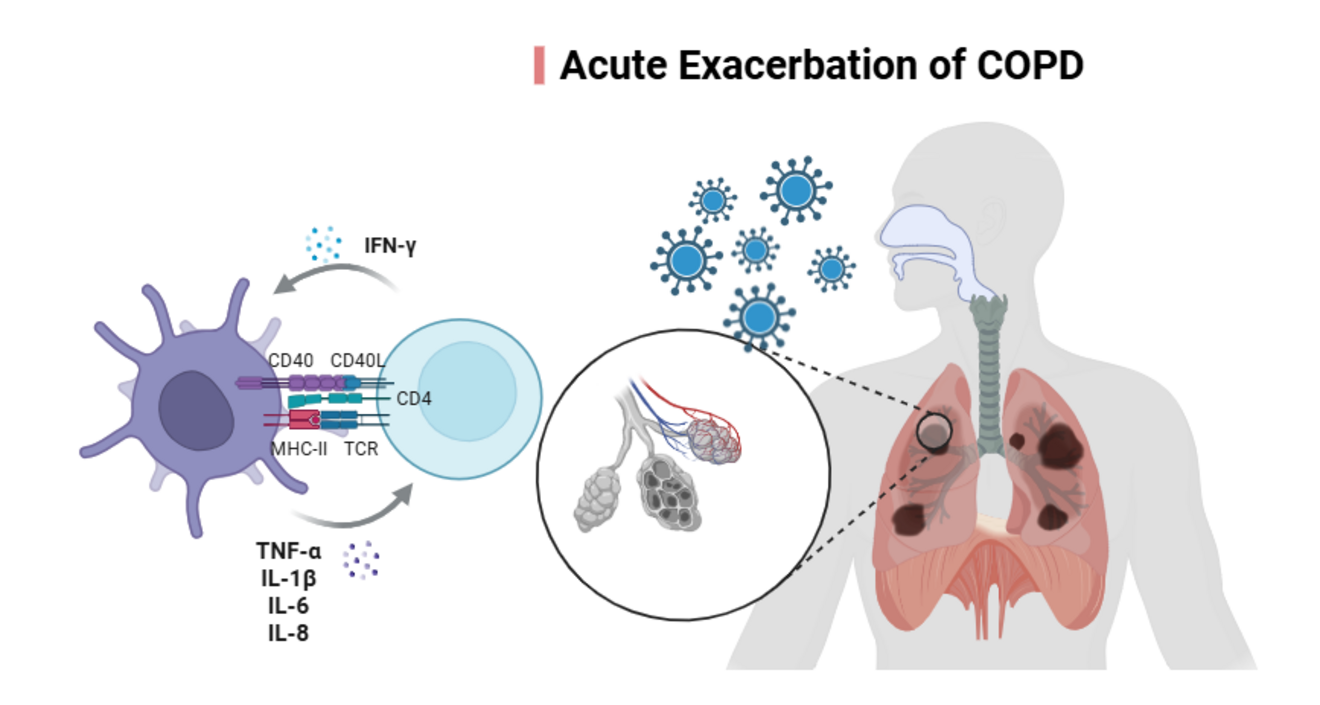
An observational study to identify potential predictors of an acute exacerbation in patients with chronic obstructive pulmonary disease (The PACE Study): Infectious triggers and prognostic biomarkers
Chronic Obstructive Pulmonary Disease (COPD) is a leading cause of morbidity and mortality worldwide. Its prevalence is estimated at 10–12% among adults over 40 years globally and 6.73% in the adult population in Germany. Acute exacerbations of COPD (AECOPD) are clinically defined as episodes of acute worsening of respiratory symptoms beyond normal daily variation, often requiring changes in treatment or hospitalization. Respiratory viruses and bacteria are detected in 60–80% of AECOPD cases. The mechanisms underlying individual susceptibility to exacerbations remain unclear, the contribution of specific pathogens to recurrent or severe episodes is not fully understood, and the ability to predict exacerbations in individual patients remains limited.
Aim: To investigate the role of viral and bacterial respiratory pathogens as potential predictors of AECOPD and to correlate pathogen presence with systemic inflammatory biomarkers measured in plasma.
Project 3
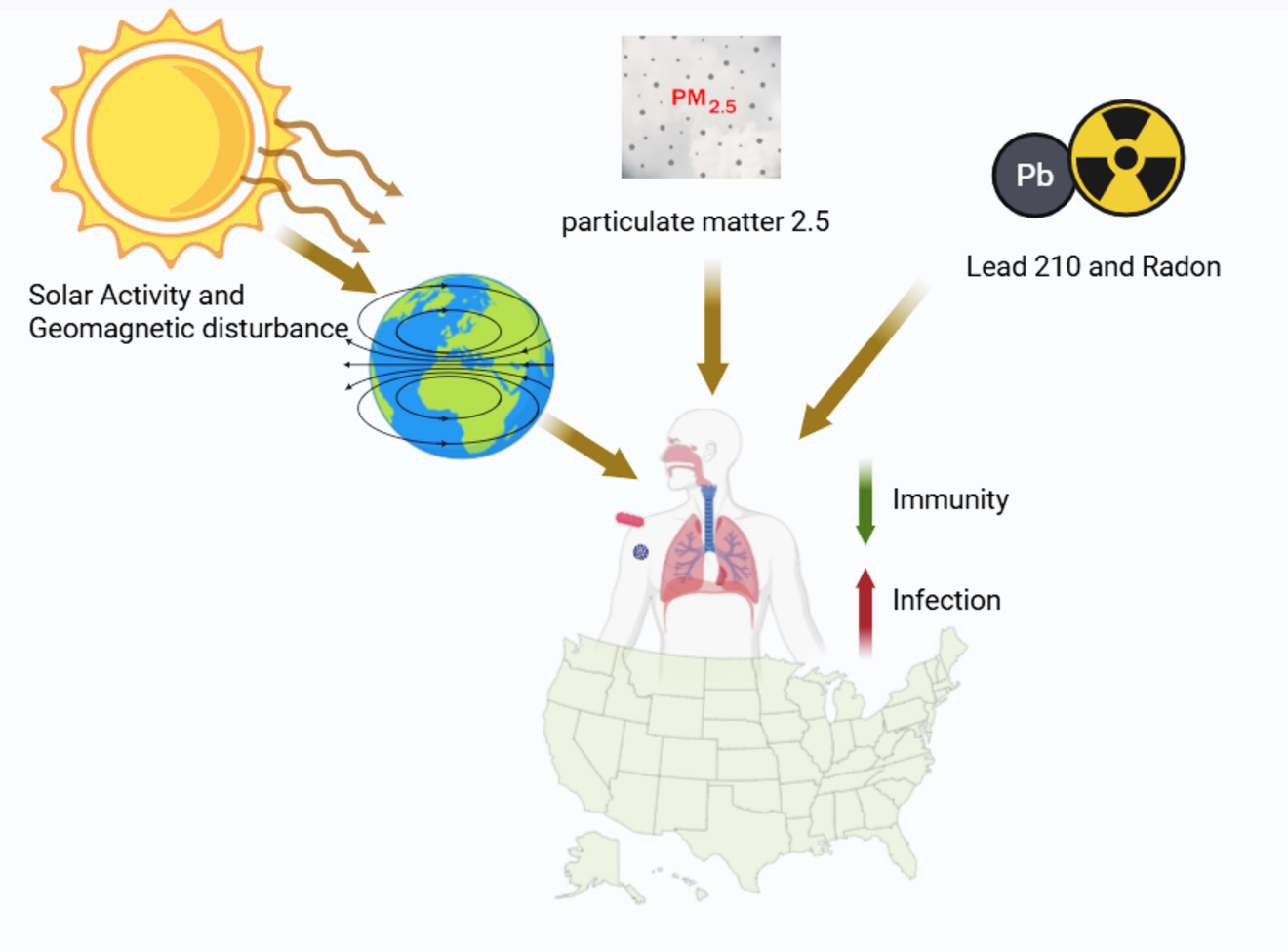
Influence of environmental factors (solar activity, outdoor radon, lead, PM2.5) on immunological biomarkers and disorders.
This project investigates how external environmental factors influence immune and inflammatory responses in humans. It focuses on exposures such as solar activity, outdoor radon, lead, and particulate matter (PM2.5), which may trigger or exacerbate systemic inflammation and modulate immune function.
We examine the effects of these environmental exposures on mortality associated with various immune-mediated diseases, as well as on immunological markers, including cytokines, C-reactive protein (CRP), and immunoglobulin E (IgE).
Overall, the findings are expected to enhance our understanding of how environmental factors contribute to both communicable and non-communicable diseases, and to inform the development of targeted prevention strategies.
Project 4
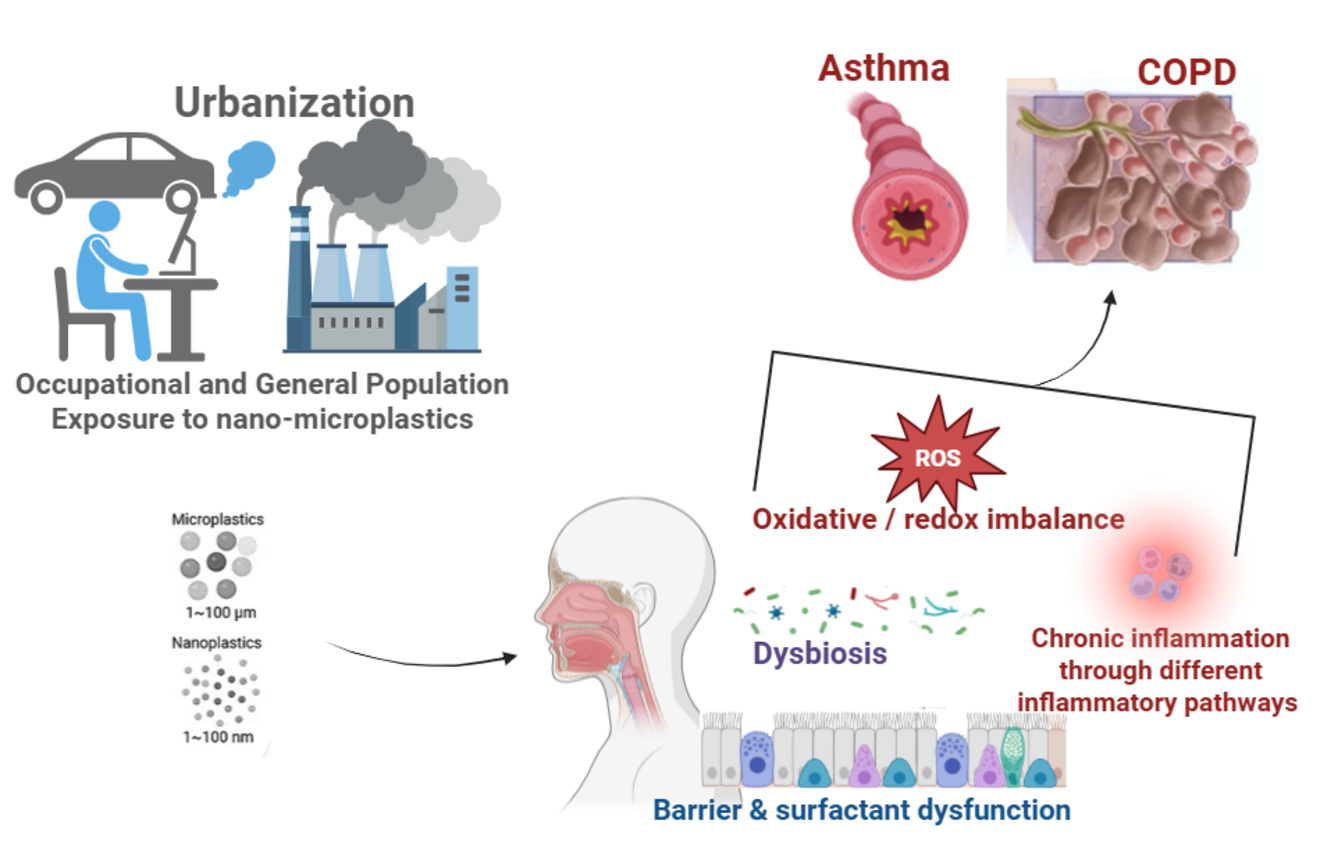
The Effects of Microplastics and Nanoplastics (MNPs) on Chronic Airway Inflammation
Exposure to airborne micro- and nanoplastics (MNPs) is increasingly recognized as a potential contributor to chronic airway inflammation and respiratory diseases such as asthma and chronic obstructive pulmonary disease (COPD). Emerging experimental evidence suggests that inhaled MNPs can damage airway epithelium and disrupt immune homeostasis through multiple mechanisms, including oxidative stress, mitochondrial dysfunction, epithelial barrier impairment, and activation of inflammatory pathways. In addition, MNPs may alter the respiratory microbiota, promoting dysbiosis and amplifying inflammatory and epithelial dysfunction.
Project 5
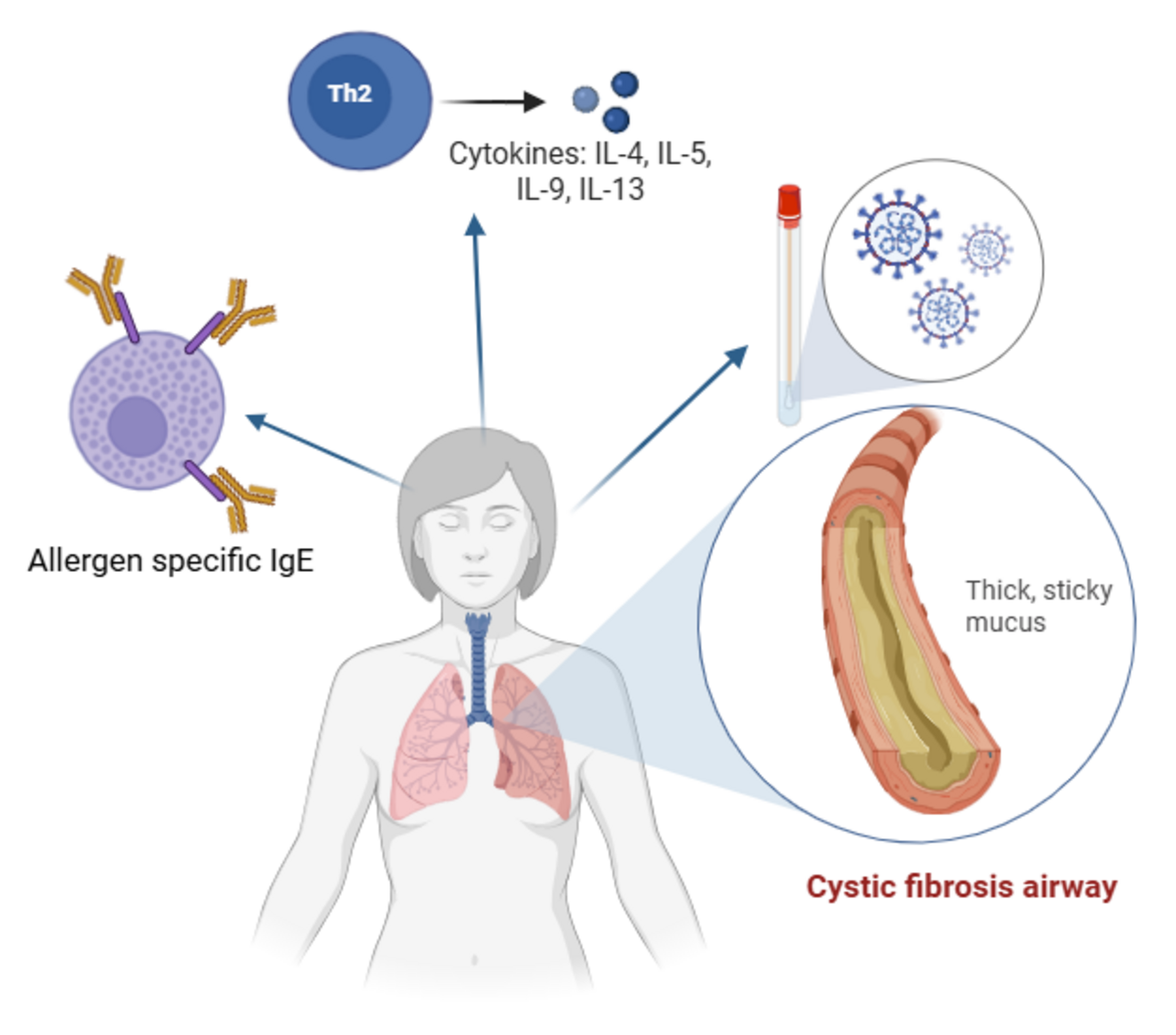
Respiratory Infections and Allergen Sensitization in Disease Morbidity and Pulmonary Exacerbations in Cystic Fibrosis and Primary Ciliary Dyskinesia: The CARMUCI Pathogens and Allergy Study
People with cystic fibrosis (pwCF) and primary ciliary dyskinesia (pwPCD) are highly susceptible to respiratory infections and exacerbations due to impaired mucociliary clearance (MCC). The contribution of viral and bacterial infections, as well as allergen sensitization, to disease morbidity and worsening in these populations remains incompletely understood.
The CARMUCI-Pathogens and Allergy study is a prospective observational study leveraging the existing CARMUCI cohort in Cyprus. We aim to assess the role of respiratory pathogens and allergen-specific immune responses in pwCF and pwPCD. Nasopharyngeal and sputum samples will be analyzed using multiplex PCR to detect viral and bacterial respiratory infections. Concurrently, serum allergen-specific IgE will be measured to identify sensitization patterns. Clinical outcomes including pulmonary exacerbations, lung function (spirometry and lung clearance index), cytokines and inflammatory biomarkers, and health-related quality of life will be monitored longitudinally. Statistical analyses will explore associations between infection burden, allergen sensitization, and disease severity.
Project 6
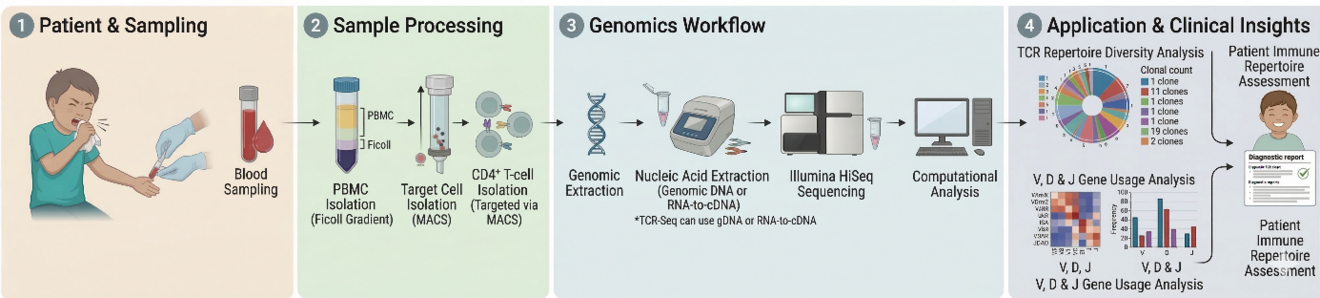
TCR repertoire analysis in childhood asthma
Asthma in early childhood represents one of the most prevalent chronic diseases in Germany, often manifesting before the age of five. Despite its frequency, reliable diagnostic biomarkers to predict disease persistence, severity, or subtype remain unavailable, as conventional lung function testing is feasible only at school age. Recent evidence indicates that adaptive immune imprinting, especially T-cell receptor (TCR) diversity, may be crucial in the pathophysiology of asthma. Comprehending the TCR repertoire may elucidate the mechanisms by which immunorecognition patterns influence various asthma phenotypes and the progression of the disease. Aim: The objective of this study is to delineate the CD4+ T-cell receptor repertoire in children suffering from asthma and recurrent wheezing, to identify immunological markers that differentiate between patients exhibiting transient symptoms and those progressing to persistent asthma. A secondary objective is to compare TCR profiles between allergic and infection-associated asthma in school-aged children to investigate the immunogenetic differences that underlie these phenotypes. Methods: Peripheral blood samples from children enrolled in the KIRA study were collected and will be processed to isolate CD4+ T cells. Genomic DNA will be extracted and analyzed using multiplex PCR targeting Vβ and Jβ segments of the TCR, followed by high-throughput sequencing on the Illumina HiSeq platform. Bioinformatic analysis will evaluate repertoire diversity, V/J usage, CDR3 length distribution, and identification of disease-associated-cell clones. Significance of the study: By establishing a connection between TCR repertoire diversity and clinical phenotypes, this study will provide novel insights into the immunological context of childhood asthma. Identification of repertoire-based biomarkers could aid with patient stratification, early disease course prediction, and the development of precision immunomodulatory treatments for childhood asthma.
Project 7
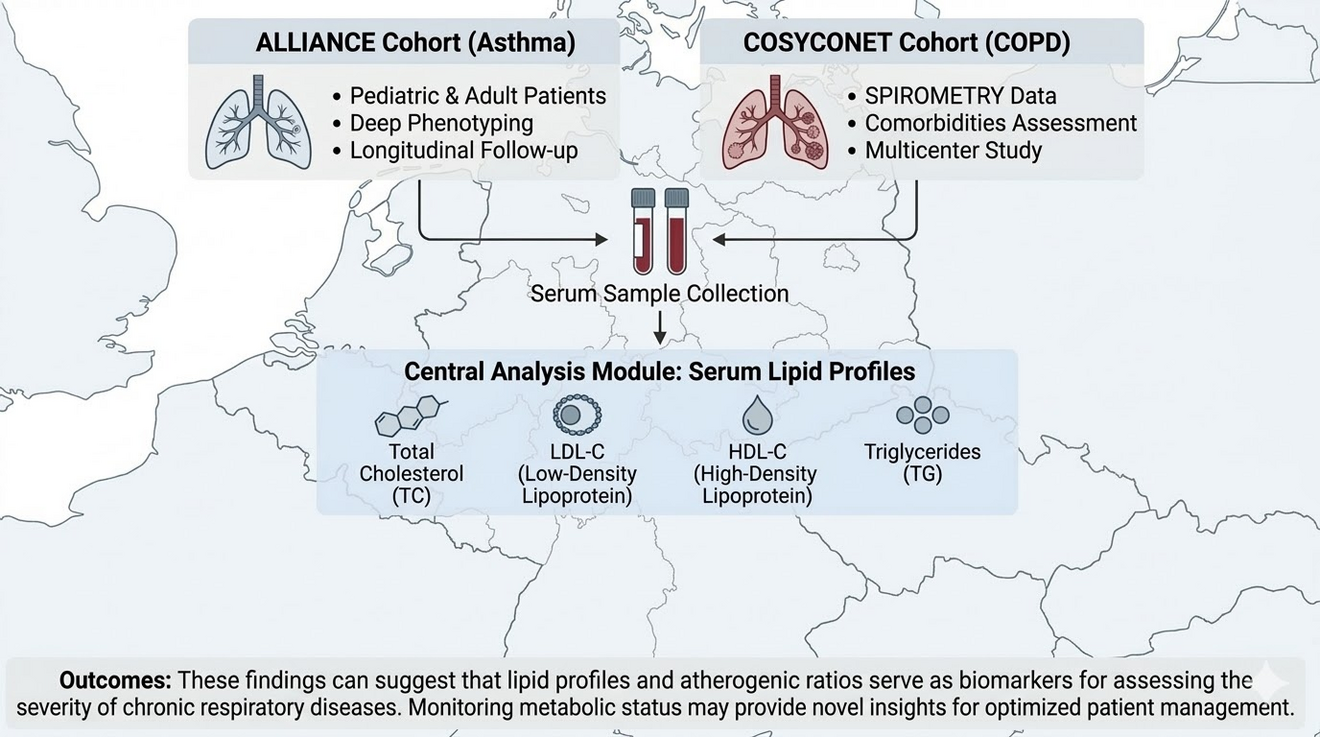
Investigation of dyslipidemia patterns and serum lipid profiles in patients with Asthma and COPD in ALLIANCE and COSYCONET Cohorts
Chronic inflammation disrupts lipid metabolism and contributes to the progression of chronic respiratory disease. However, the exact relationships between serum lipid profiles, lung function, disease severity, and inflammatory phenotypes in asthma and chronic obstructive pulmonary disease (COPD) remain incompletely understood. Objectives: The primary objective of this study was to identify specific patterns of dyslipidemia associated with asthma and COP severity and to evaluate serum lipid profiles as potential biomarkers for disease control and inflammatory endotypes. Data were analyzed from two large-scale prospective German cohorts: ALLIANCE (Asthma) and COSYCONET (COPD). Serum lipid parameters (TC, LDL, HDL, TG) and atherogenic ratios were assessed. Multivariable linear and logistic regression models, adjusted for age, sex,BMI, and smoking history, were employed to evaluate the associations between lipid profiles and clinical outcomes, including lung function (FEV1, FVC) and disease severity. Overall, these findings suggest that lipid profiles and atherogenic ratios serve as biomarkers for assessing the severity of chronic respiratory diseases. Monitoring metabolic status may provide novel insights for optimized patient management.
Project 8
Effect of Exposure to Radon decay products on COPD
Chronic Obstructive Pulmonary Disease (COPD) is a leading cause of global mortality, characterized by chronic airway inflammation and impaired lung function. While tobacco smoking remains the primary risk factor, environmental triggers like radon gas and its decay products, such as Pb-210, contribute significantly to the disease burden. These radioactive elements emit alpha particles that damage pulmonary epithelial cells, yet the specific link between radon and COPD remains scientifically inconsistent compared to its well-established role in lung cancer. Some studies suggest a synergistic risk with heavy smoking, while others show no clear association, highlighting a need for more robust population-based data. Consequently, this study aims to examine the relationship between environmental radon exposure and COPD prevalence and mortality. By clarifying these conflicting results, we hope to provide novel insights into the environmental determinants of chronic respiratory health.
Project 9
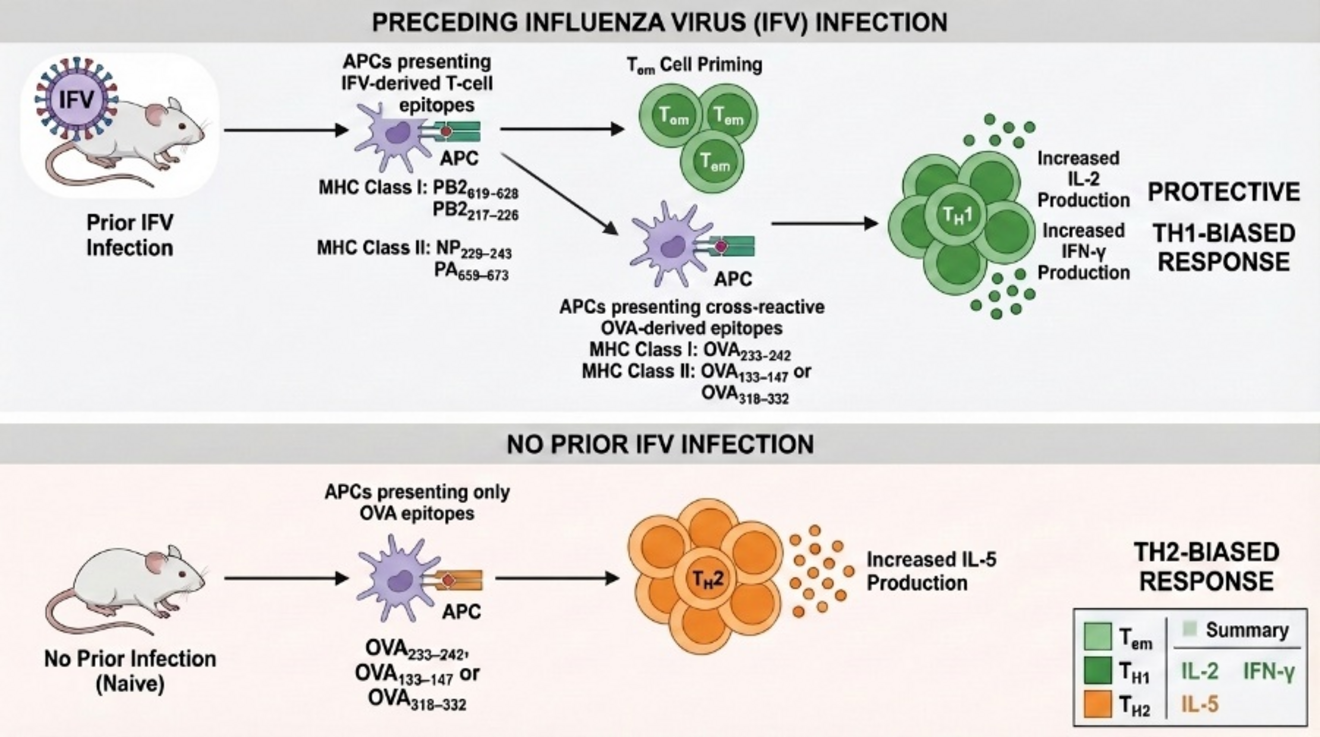
Cross-reactive T-cell immunity between respiratory viruses and allergens
Respiratory viral infections can induce long-lasting immune responses that influence susceptibility to allergic diseases. In this project, we investigate whether virus-specific T cells recognize structurally related allergen-derived epitopes, leading to cross-reactive immune responses. Using immunoinformatics-based epitope prediction, peptide immunization strategies, and functional T-cell assays, we study whether virus-derived peptides can induce protective CD8⁺ T-cell responses that modulate allergic airway inflammation. These studies aim to identify mechanisms through which viral immunity may influence allergic disease development and potentially inform future preventive immunization strategies.
Project 10
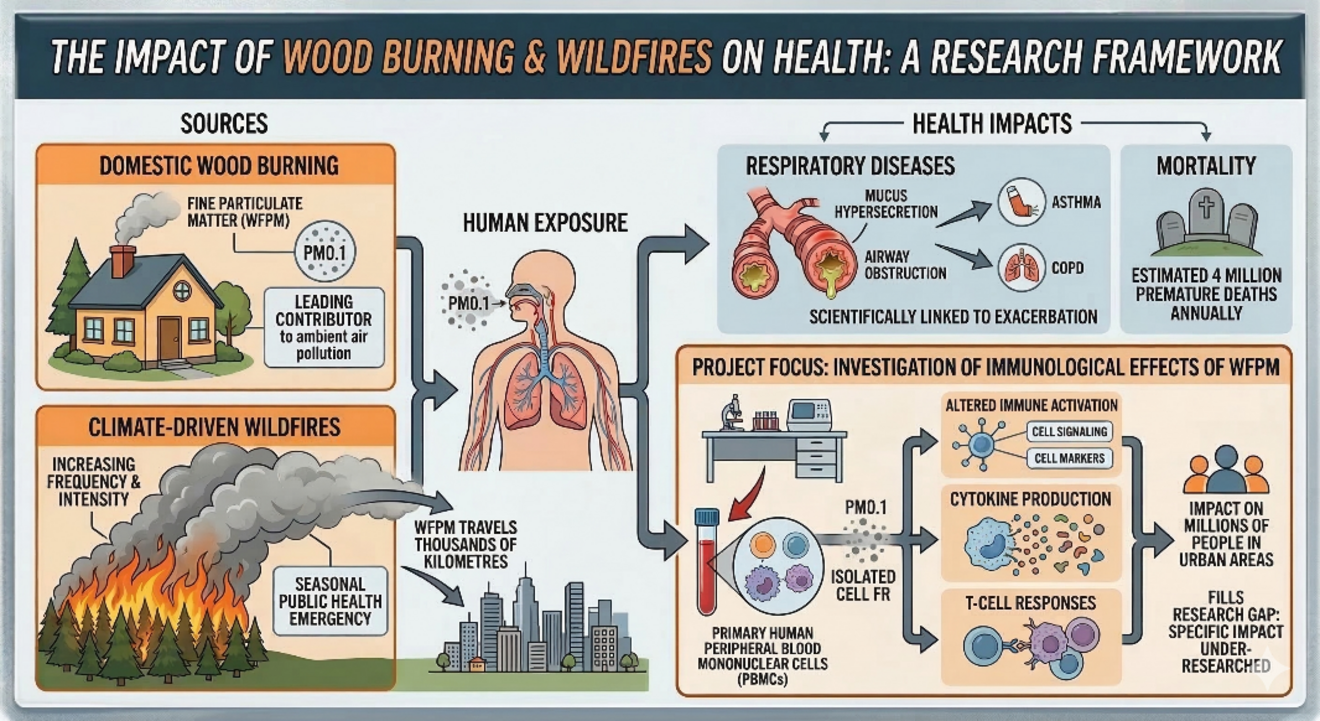
Immune responses to wood smoke fine particulate matter (WFPM)
Air pollution represents an important environmental risk factor for respiratory diseases, including asthma and chronic airway inflammation. In this project, we investigate the immunological effects of wood smoke fine particulate matter (WFPM) on human immune cells. Using primary human peripheral blood mononuclear cells (PBMCs), we examine how exposure to WFPM alters immune activation, cytokine production, and T-cell responses. Special attention is given to the interaction between environmental particulate exposure and antiviral immune responses. Understanding how airborne particulate matter shapes immune function may provide important insights into how environmental pollution contributes to respiratory disease susceptibility and immune dysregulation.
Project 11
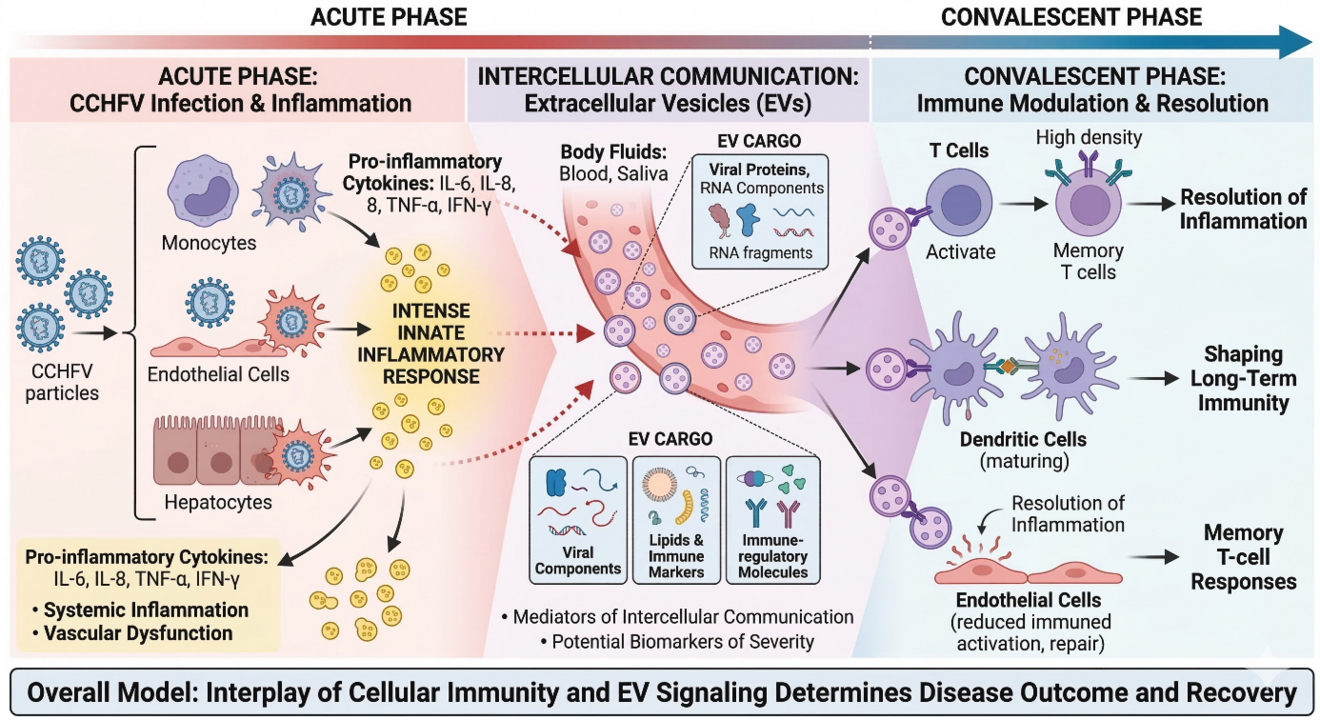
Cellular and extracellular immune responses in Crimean-Congo hemorrhagic fever virus (CCHFV) infection
Crimean-Congo hemorrhagic fever virus (CCHFV) is a highly pathogenic tick-borne virus with case fatality rates reaching up to 30%. Despite its clinical relevance and expanding geographic distribution, the immune mechanisms determining disease severity and recovery remain poorly understood. This project investigates how both cellular immunity and extracellular vesicle (EV)-mediated signaling contribute to immune regulation during and after CCHFV infection.
Using clinical samples collected from CCHFV patients at different stages of infection and recovery, we characterize antiviral immune responses by integrating serology, cytokine profiling, and extensive immunophenotyping of T-cell subsets. In parallel, we analyze extracellular vesicles isolated from blood and saliva to determine their molecular composition, cellular origin, and antiviral activity. Special emphasis is placed on the interaction between EVs and immune cells, as EVs can transfer viral or host-derived signals that influence inflammation, immune activation, and viral control.
By combining high-dimensional immune profiling, functional immune assays, and extracellular vesicle characterization, this project aims to identify immunological biomarkers associated with disease severity and recovery. Understanding how cellular and extracellular immune mechanisms interact during CCHFV infection may provide new opportunities for prognostic biomarker development and future therapeutic strategies.
Project 12
Establishment of an accessible, digitally-based regional care infrastructure for
the appropriate diagnosis and treatment of post-COVID (PROGRESS – Biomarkers Module)
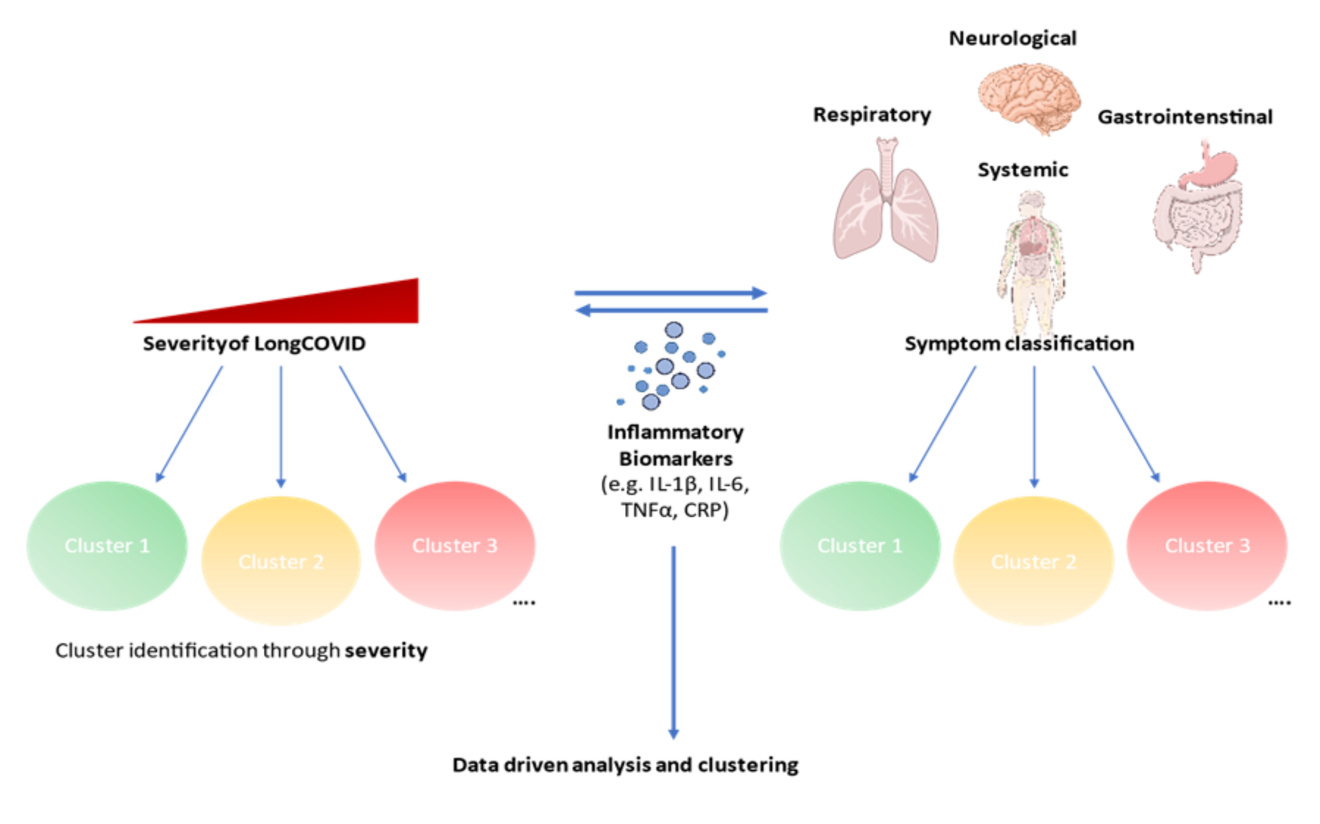
Post-COVID or Long COVID poses a particular challenge to the healthcare system due to its heterogeneous presentation. The condition places a significant burden on the healthcare system (Cutler 2022). Various studies have already attempted to classify post-COVID into clusters, with one of the most common methods being clustering based on symptoms (Wang et al. 2026). Multi-omics studies in smaller patient populations have found that post-COVID is a heterogeneous disease at the transcriptomic and proteomic levels (Lin et al. 2024). We predict that different post-COVID clusters are associated with varying levels of elevated biomarkers. We are investigating whether and how biomarkers differ in post-COVID patients and to what extent they evolve over the course of the disease. Based on this, we aim to identify markers that can be used to predict the course of post-COVID.
Project 13
Establishment of an accessible, digital-based regional care infrastructure for the appropriate diagnosis and treatment of post-COVID conditions (PROGRESS – Gender and Pregnancy Module), Philipps University of Marburg
While there is evidence that pregnant women are more susceptible to acute illness during SARS-CoV-2 infection, little is known about the risk of post-COVID in the context of pregnancy. However, preliminary data suggest an increased risk of preeclampsia in post-COVID patients. The existing data remain controversial, however, as both an increased risk (Backes et al. 2024) and a reduced risk of post-COVID have been observed in women who experienced a SARS-CoV-2 infection during pregnancy (Backes et al. 2024). This topic remains controversial, as other publications show that pregnant women have a lower risk of developing post-COVID disease (Zang et al. 2025).
Since sex and pregnancy hormones can influence the immune system, relevant laboratory parameters (e.g., IGF-1) and hormonal status (e.g., estrogen, progesterone, IGF-1, LH, testosterone, and prolactin) are assessed in all patients during pregnancy. Furthermore, the module will address gender-specific differences between women and men regarding post-COVID, as studies have found that women have an increased risk of developing post-COVID (Wang et al. 2024). By broadly covering various biomarkers associated with the course of pregnancy, we aim to investigate whether and to what extent complications can be predicted. Furthermore, the influence of hormonal differences among post-COVID patients will be examined. We will analyze how these differences vary over the course of the disease and to what extent the specific differences are associated with post-COVID.
Project 14
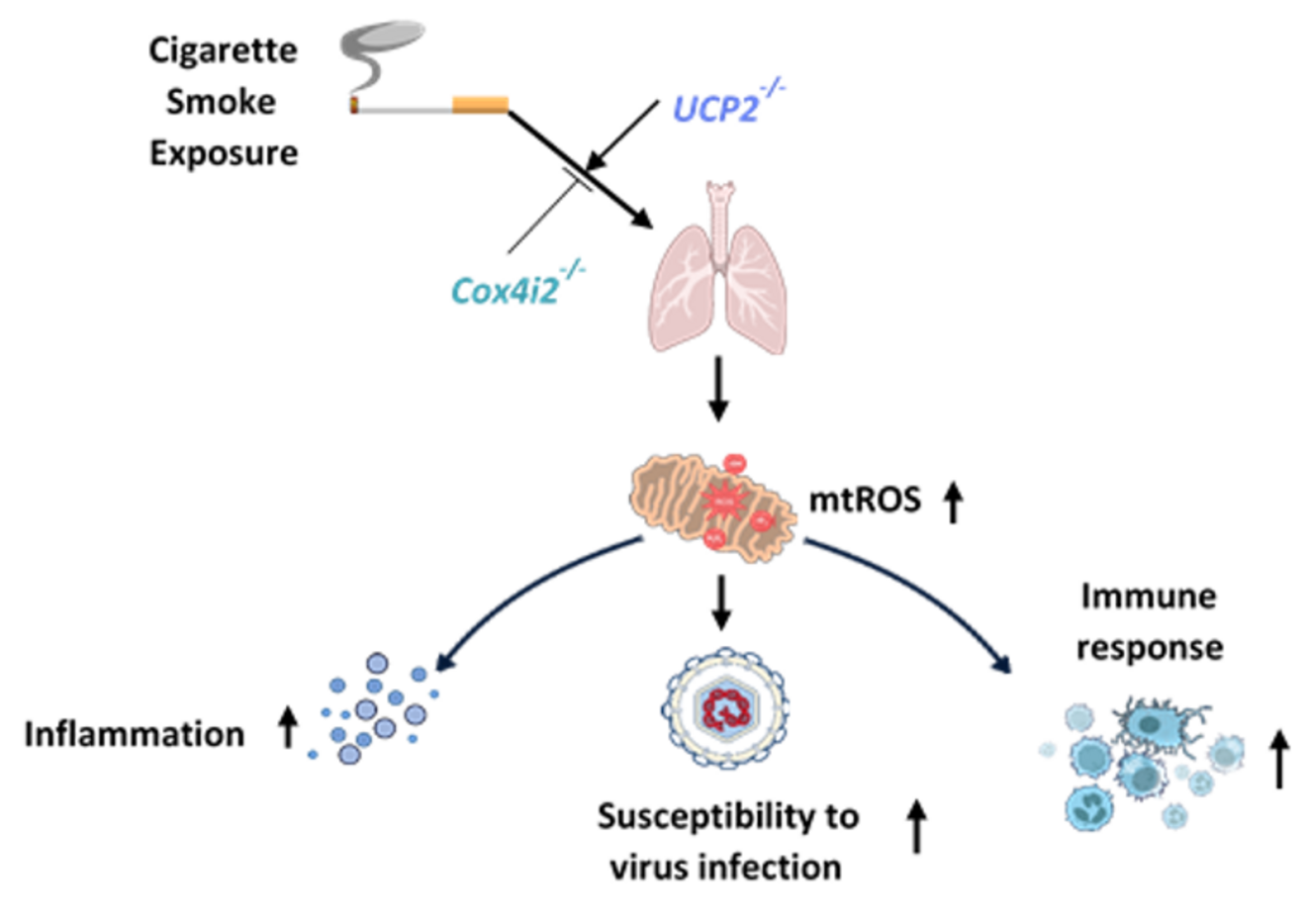
Mitochondrial T-Cell reprogramming in virus-induced COPD exacerbation/emphysema progression
Chronic obstructive pulmonary disease (COPD) ranks among the top 3 global causes of death. Virus-induced exacerbations are heavily responsible for the prominent morbidity, mortality and associated health care costs. COPD patients suffer from increased susceptibility to- and decreased clearance from- respiratory viruses, as well as hyperinflammation in response to associated infections, which promotes lung injury and permanent lung remodelling. Enhanced susceptibility and immunopathology have been associated with distorted innate and adaptive immune responses, in particular with the crosstalk of macrophages and bronchial epithelial cells (BEC) with T cells. Mitochondria may be critically involved in this intercellular communication. We hypothesize that (i) chronic smoke exposure-induced mitochondrial dysfunction (including aberrant mtROS, MAVS) causes impaired T cell function (activation, proliferation, differentiation), which enhances susceptibility to respiratory virus infections in COPD, (ii) recurrent virus infections excessively activate mitochondrial signaling, promote hyperinflammation and T cell exhaustion, and ultimately emphysema and (iii) targeting mitochondrial reprogramming of T cells and mtROS/MAVS-signaling in macrophages/BEC will allow augmentation of antiviral immune responses and prevent disease exacerbations and progression of emphysema.
Publications
- Skevaki C, Nadeau KC, Rothenberg ME, et al. Impact of climate change on immune responses and barrier defense. J Allergy Clin Immunol. 2024;153(5):1194-1205. doi:10.1016/j.jaci.2024.01.016
- Balz K, Grange M, Pegel U, et al. A novel mass cytometry protocol optimized for immunophenotyping of low-frequency antigen-specific T cells. Front Cell Infect Microbiol. 2024;13:1336489. Published 2024 Jan 15. doi:10.3389/fcimb.2023.1336489
- Skevaki C, Chinthrajah RS, Fomina D, et al. Comorbidity defines asthmatic patients' risk of COVID-19 hospitalization: A global perspective. J Allergy Clin Immunol. 2023;151(1):110-117. doi:10.1016/j.jaci.2022.09.039
- Fragkou PC, De Angelis G, Menchinelli G, et al. ESCMID COVID-19 guidelines: diagnostic testing for SARS-CoV-2. Clin Microbiol Infect. 2022;28(6):812-822. doi:10.1016/j.cmi.2022.02.011
- Chang SE, Feng A, Meng W, et al. New-onset IgG autoantibodies in hospitalized patients with COVID-19. Nat Commun. 2021;12(1):5417. Published 2021 Sep 14. doi:10.1038/s41467-021-25509-3
- Skevaki C, Tafo P, Eiringhaus K, et al. Allergen extract- and component-based diagnostics in children of the ALLIANCE asthma cohort. Clin Exp Allergy. 2021;51(10):1331-1345. doi:10.1111/cea.13964
- Skevaki C, Karsonova A, Karaulov A, et al. SARS-CoV-2 infection and COVID-19 in asthmatics: a complex relationship. Nat Rev Immunol. 2021;21(4):202-203. doi:10.1038/s41577-021-00516-z
- Balz K, Kaushik A, Chen M, et al. Homologies between SARS-CoV-2 and allergen proteins may direct T cell-mediated heterologous immune responses. Sci Rep. 2021;11(1):4792. Published 2021 Feb 26. doi:10.1038/s41598-021-84320-8
- Renz H, Skevaki C. Early life microbial exposures and allergy risks: opportunities for prevention. Nat Rev Immunol. 2021;21(3):177-191. doi:10.1038/s41577-020-00420-y
- Skevaki C, Hudemann C, Matrosovich M, et al. Influenza-derived peptides cross-react with allergens and provide asthma protection. J Allergy Clin Immunol. 2018;142(3):804-814. doi:10.1016/j.jaci.2017.07.056
Other publications by Prof. Dr. med. Chrysanti Skevaki
Research Group Leader

Univ.-Prof. Dr. Chrysanthi Skevaki
Research Group Members
| Sara Völkel | Thilo Berger |
| Zain Karamya | Ariana Younes |
| Rim Harfouch | Uta Pegel |
| Katharina Parzefall | Silva Herzog |
| Katharina Bremerich | Ercan Arslan |
| Lea Nadolny | Alexander-Tobias Hensel |
| Lisa Zheng | Tom Pink |
| Florian Schmidt | Svenja Hartmann |
| Samuel Asri | Aaron Meilinger |
| Paula Roder | Uyen Trinh |
| Metin Soylu | Maryam Sammari |
| Christin Fidera | Frederike Schwarzer |
| Javad Haratizadah |
Collaboration partner
- Prof. Dr. Kari Christine Nadeau, Harvard University, Allergy, Extreme Weather, and Exposomics Lab, Boston, USA
- Prof. Dr. Holden Maecker, Stanford University, Human Immune Monitoring Center (HIMC), California, USA
- Dr. Hervé Luche, CIPHE, Research platfrom, Marseilles, France
- Prof. Dr. Stefan Bauer, Philipps University Marburg, Institute of Immunology, Marburg, Germany
- Dr. Jochen Wilhelm, Justus-Liebig University, Genomics and Bioinformatics Lab, Gießen, Germany
- Prof. Dr. Marek Bartkuhns, Justus-Liebig University, Biomedical Informatics and Systems Medicine Science Unit, Gießen, Germany
- Dr. Petros Koutrakis, Harvard University, Department of Environmental Health, Boston, USA
- PhD Dr. Philipp Demokritou, Rutgers University, New Brunswick, USA
- PD Dr. Paolo Matricardi, Charite, Berlin, Germany
- Prof. Dr. Rudolf Valenta, Vienna Medical University, Working Group Allergology and Immunology, Vienna, Austria
- Prof. Dr. Susanne Herold, Justus-Liebig University, Internal Medicine, Infectious Diseases & Pulmonary Research, Gießen, Germany
- Prof. Dr. Natascha Sommer, Justus-Liebig University, Mitochondrial physiology and pathology in the lung, Gießen, Germany
- Prof. Dr. Paul J. Utz, Stanford University, Immunology & Rheumatology, California, USA
- Prof. Dr. Bianca Schaub, Ludwig-Maximilian University Munich, Schaub Lab, Munich, Germany
- Prof. Dr. Erika von Mutius, Ludwig-Maximilian University, Institute of Asthma and Allergy Prevention, Munich, Germany
- Prof. Dr. Gesine Hansen, Medical University of Hannover, Allergy and tolerance, Hannover, Germany
- Prof. Dr. Anna-Maria Dittrich, Medical University of Hannover, Inflammatory lung diseases in childhood, Hannover, Germany
- Prof. Dr. Markus Weckmann, University of Lübeck, Center for Infectious Diseases and Inflammation Research, Lübeck, Germany
- Prof. Dr. Rembert Koczulla, Philipps University Marburg, Marburg, Germany
- Prof. Dr. Blandina Theophil Mmbaga, Kilimanjaro Christian Research Institute, Moshi, Tanzania
- Dr. Jon Sanchez & Prof. Alfonso Valencia, Supercomputing Center, Computational Biology, Barcelona, Spain
Funding
| Project | Time frame | Provider |
| Establishment of a barrier-free, digital-based regional care structure for the adequate diagnosis and treatment of post-COVID | 2024 - 2027 | BMG/BVA |
| Asthma and Allergy (FKZ 82DZL005C2) | 2024 - 2027 | DZL (BMBF) |
| Asthma and Allergy (FKZ 82DZL005A2) | 2021 - 2023 | DZL (BMBF) |
| RNA viruses induce heterologous immune protection from atopy and asthma (SFB 1021) | 2021 - 2024 | DFG |
| RNA viruses induce heterologous immunity against environmental allergens (SFB 1021) | 2017 - 2021 | DFG |
| Mitochondrial T Cell Reprogramming in virus-induced COPD Exacerbation/ Emphysema Progression (KFO 309) (SK317/2-1) | 2020 - 2024 | DFG |
| The development of the antibody repertoire against rhinoviruses and house dust mites in relation to the onset and persistence of bronchial asthma: a high-resolution analysis in humans and mice (SK317/1-1) | 2019 - 2024 | DFG |
| A systems vaccinology approach to assess influenza vaccine immunogenicity in asthma and COPD patients (P-Nr.: 17/2017) | 2017 - 2025 | UKGM, Rhön Klinikum |
| Deep Asthma endotyping among adult Maasai and urban Kilimanjaro, Tanzania (DEAPMARK) Pretest Study: Feasibility and quality development of biomaterials | 2020 - 2023 | Stiftung Pathobiochemie |